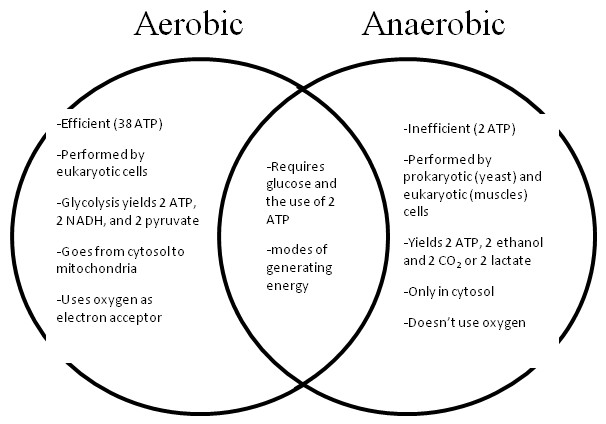
Compare and contrast Anaerobic cellular respiration and Aerobic cellular respiration. In your answer, address glycolysis, the citric acid cycle, oxidative phosphorylation, lactic acid fermentation, alcohol fermentation, NADH, FADH2 and ATP. At the most basic level, the difference between Anaerobic and Aerobic respiration is that Aerobic respiration uses O2 and Anaerobic doesn’t. Aerobic also produces much more ATP than Anaerobic does (28-38 vs. 6ish). Both types of respiration include glycolysis in the cytosol and produce 2ATP and NADH. Only aerobic respiration has the citric acid cycle (produces 2 ATP and FADH2) and oxidative phosphorylation (produces 26-28 ATP) both of which occur in the mitochondria. Lactic acid and alcohol fermentation are both used in anaerobic respiration. In lactic acid fermentation, pyruvate reduced by NADH, forms lactate. No CO2 is released (2 ATP produced). With alcohol fermentation, pyruvate is reduced to ethanol in 2 steps; 1. CO2 released from pyruvate and 2. acetaldehyde reduced to ethanol.
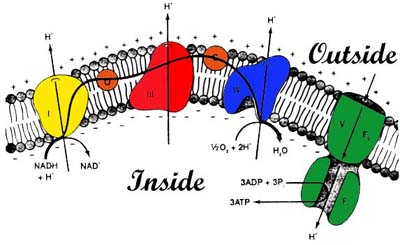
Explain why the disruption of chemiosmosis and the proton motive force can be detrimental to eukaryotic organisms. Provide a real life example. When is chemiosmosis (energy coupling mechanism that uses energy stored in the form of a H+ gradient across a membrane) and the proton motive force (the gradient of hydrogen ions) are disrupted a eukaryotic organism can not produce ATP and you would see mass cell death and ultimately the death of the organism because no energy is being produced. A real life example is cyanide poisoning. Cyanide damaged the mitochondria and prohibited the production of APT and killed people.
Compare and contrast obligate and facultative anaerobes. Obligate anaerobes and facultative anaerobes carry out fermentation. But only obligate anaerobes carry out anaerobic respiration, but cannot survive in the presence of O2. On the other hand, facultative anaerobes can carry out cellular respiration.
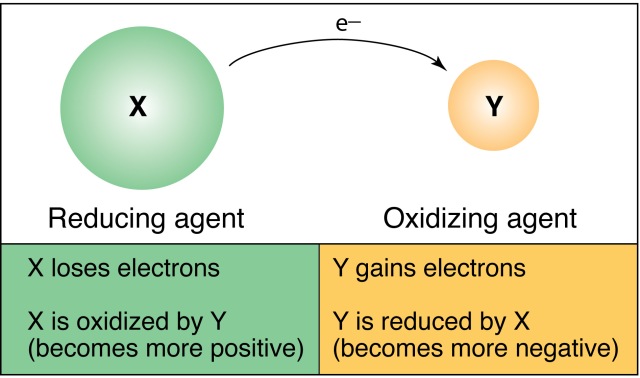
In the following redox reaction, identify which molecules have been oxidized and reduced. Also identify the reducing agents and the oxidizing agents.
C6H12O6 + 6O2 → 6CO2 + 6H2O + Energy
Carbon is oxidized due to the conversion from C6 → 6CO2 (looses electrons) this makes carbon the reducing agent.
Oxygen is reduced by taking electrons from the less electronegative Carbon (O6 → 6CO2), making oxygen the oxidizing agent.